But we have to remember that as we move from left to right, the electrons are added to the same energy level or to lower energy levels (as the d and f shells fill up), closer to the nucleus. This may seem counter intuitive-after all, each new element to the right has more electrons, which should take up more room. In general, atomic radii decrease from left to right across the periodic table. If you want to compare them to something, they are more like the gas planets Jupiter and Saturn, where gas density gradually increases as we move to the center of the planet, but where there is no planetary surface as there is on earth or Venus.

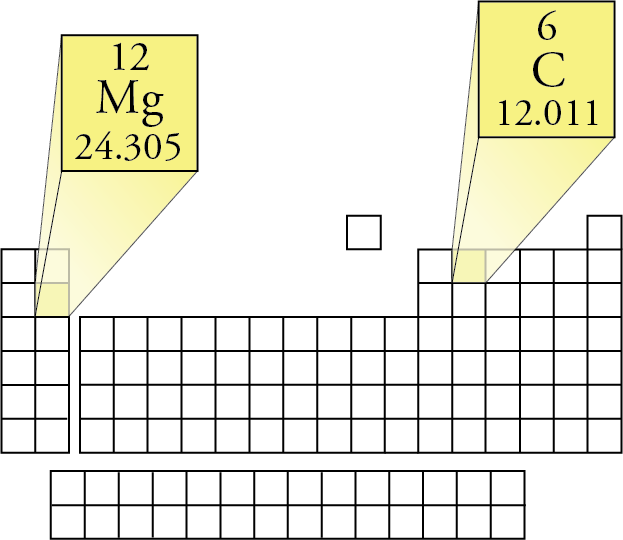
These are estimates-the electron shells have no solid surfaces. How does the radius of the atom when potassium K loses an electron? When carbon loses an electron? Is there a trend in the radius change? Notice that we assume no vacuum between the atoms.Ĭompare the size of neutral atoms (red) and ions (green) in this periodic table arrangement of radius information. For example, in diamond, the carbon nuclei are 154 picometers apart, so the diameter of each carbon atom is estimated to be half that, or 77 pm. Sizes can be estimated by finding the distance between centers of atoms in pure substances of the element (using electron microscopes to examine the surface of the sample). So mass should increase across periodic table rows from left to right and down periodic table columns experimental evidence supports this prediction.Ītomic size does not follow such simple rules. We know that mass increases as the atomic number increases, and depends primarily on the number of protons and neutrons in the atoms. We've discussed mass at length by doing molar mass conversions. Atomic propertiesĪtomic properties include size and mass. Now we look at other "trends" in the table for atomic size, ionization energy, electron affinity, ion sizes, and chemical reaction properties. Last time, we looked at how the periodic table can give us clues to the electron configuration of different elements in their ground state. Optional Website Reading Periodic Table Position and Electron Configuration.Ionization Energy and Electron Affinity.Periodic Table Position and Electron Configuration.CO 2 has one carbon atom and two oxygen atoms.Oxygen (O) has an atomic mass of about 16.00 amu.Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Find atomic masses: look up the atomic masses of each element present in the compound.For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. Identify the compound: write down the chemical formula of the compound.One mole contains exactly 6.022 ×10 23 particles (Avogadro's number) Mole is a standard scientific unit for measuring large quantities of very small entities such as atoms and molecules.Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol.(1 u is equal to 1/12 the mass of one atom of carbon-12) Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u).To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations: Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) Functional groups: D, Ph, Me, Et, Bu, AcAc, For, Tos, Bz, TMS, tBu, Bzl, Bn, DmgĮxamples of molar mass computations: NaCl, Ca(OH)2, K4, CuSO4*5H2O, nitric acid, potassium permanganate, ethanol, fructose, caffeine, water.Capitalize the first letter in chemical symbol and use lower case for the remaining letters: Ca, Fe, Mg, Mn, S, O, H, C, N, Na, K, Cl, Al. Computing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'.
